One of Medicine's Most Underrated Heroes
The most common sight in any hospital room is also the most critical. What sterile injectables actually do—and why the quality behind them is never just a manufacturing detail
1/31/20262 min read
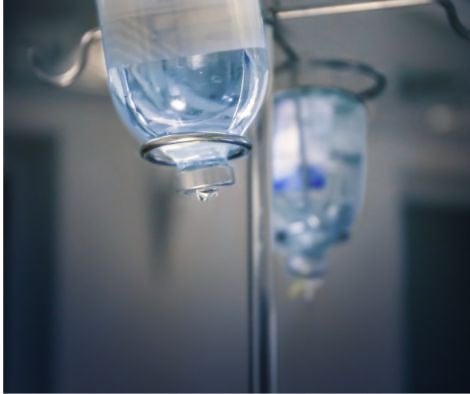
You've seen it in every hospital room. A clear bag hanging from a metal pole, a thin tube running into someone's arm, liquid moving slowly and steadily into the bloodstream. It's so common it's become wallpaper. Background noise in the visual grammar of illness and recovery.
But that unremarkable bag might be the most important thing in the room.
Injectables — IV fluids, sterile solutions, intravenous medications — are the backbone of acute medical care. When everything else fails or isn't fast enough, injectables work. That's not a marketing claim. That's basic pharmacology.
Why Injectables Matter
When a patient can't swallow, injectables keep them alive. When a drug needs to reach the bloodstream immediately — in sepsis, in cardiac arrest, in severe dehydration — the oral route is too slow. The digestive system takes time. Time a critically ill patient doesn't have.
Injectables bypass all of that. Intravenous delivery means 100% bioavailability — the full dose reaches the bloodstream, no absorption variables, no first-pass metabolism through the liver diluting the effect. What you give is what the patient gets, precisely and immediately.
This is why sterile injectables are standard of care in ICUs, operating rooms, and emergency departments worldwide. Dextrose solutions restore blood sugar in hypoglycemic crises. Sodium chloride rehydrates patients who've lost dangerous amounts of fluid. Water for injection is the vehicle that carries antibiotics, chemotherapy drugs, and critical medications into the body safely.
Remove injectables from a hospital and you've essentially removed the hospital's ability to manage any serious acute condition. That's how foundational they are.
What Nobody Tells You About Quality
Here's the part that gets glossed over in most health explainers: not all injectables are created equal, and the difference is not always visible.
Sterile injectable manufacturing is one of the most regulated areas in pharmaceutical production — and for good reason. These products go directly into the bloodstream. There is no digestive system to filter out contaminants. No second chance. A compromised injectable can cause bloodstream infections, sepsis, or death.
This is why the manufacturing environment matters enormously. Grade A filling conditions. Validated sterilization processes. Rigorous quality control testing on every batch. Unbroken cold chain integrity from production to bedside.
When evaluating an injectable product or the manufacturer behind it, these are the non-negotiables:
Current Good Manufacturing Practice (cGMP) compliance. This is the regulatory floor, not the ceiling. Look for manufacturers that treat it as a baseline, not an achievement.
Transparent quality documentation. Certificates of Analysis should be readily available. Batch traceability should be standard. If a manufacturer can't tell you exactly how a product was made and tested, that's information.
Packaging integrity. The container is not cosmetic. Glass is preferred for sterile injectables because it doesn't interact with the product, doesn't leach chemicals under temperature variation, and maintains product integrity longer. The choice of packaging material is a quality decision, not just a cost one.
Track record in the market. Injectable manufacturers live and die by their reliability. Hospital pharmacies and purchasing departments have long institutional memories. A company that has supplied Philippine hospitals for decades without major quality incidents has earned that tenure.
Local matters more than you think. Philippine-owned manufacturers operating to international standards give the local healthcare system supply chain resilience. When global disruptions hit — and they do — local production capacity is what keeps hospitals running.
The IV drip hanging in that hospital room looks simple. The supply chain, the manufacturing standards, and the quality decisions behind it are anything but.
Pay attention to what's in the bag. And who made it.
Quality & Reliability
Delivering safe and effective pharmaceutical solutions.
BEST DRUG INDUSTRIES
AND PHARMA
622 Sta. Cecilia Street, San Antonio Valley 1, 1700 Parañaque City Philippines
© 2024. All rights reserved.
